I am always on the lookout for interesting, engaging, and exciting ways to teach my kids. Designing an experiment to inflate a balloon with dry ice is the perfect dry ice experiment for kids to have a ton of fun while learning about scientific topics such as changing states of matter, carbon dioxide, cloud formation, or gases in general. It’s easy to do with dry ice and water in the classroom or at home.
If you enjoy this dry ice experiment and are wondering what else to do with dry ice, be sure to check out our ebook, 8 SUPER COOL DRY ICE SCIENCE EXPERIMENTS. It is jam-packed with bubbly, hands-on, exciting dry ice projects that are suitable for learning and fun in the classroom or at home. Click the picture below to find out more!
Be sure to scroll to the end of this post where you will find a link to the popular 28 Days of STEAM series going on this month! The most brilliant bloggers on the web have come together to share their best science, technology, engineering, art, and math activities all in one place. Don’t miss it!
Getting Ready
*This post contains affiliate links. Please see our disclosure policy.
*Safety Alert* Dry ice is relatively safe, but it can be dangerous if handled incorrectly. Please see our dry ice safety FAQs for more information about safety, storage, and handling.
My kids and I love to have fun with dry ice! It is such an interesting material and there is so much to learn by doing experiments with dry ice.
We decided that we wanted to catch the carbon dioxide gas as it sublimes, or changes from a solid to a gas. Setting up a dry ice experiment where we inflate a balloon with dry ice seemed like a simple experiment for the kids and me to do.
To inflate a balloon with dry ice we gathered the following supplies:
- Bottle with a narrow neck
- Warm water
- Food coloring (optional)
- Dry ice
- Balloon
Whenever we do science experiments with dry ice we always wear gloves and eye protection, just to be on the safe side. It can’t hurt, right??
I filled the bottle about 3/4 of the way full of warm water. We added a few drops of food coloring, just for fun.
Using a hammer, I broke off several grape-sized chunks of dry ice from the block. They had to be small enough to fit through the neck of the bottle.
With our balloon close by we were ready to begin our dry ice science experiment!
Inflate a Balloon with Dry Ice
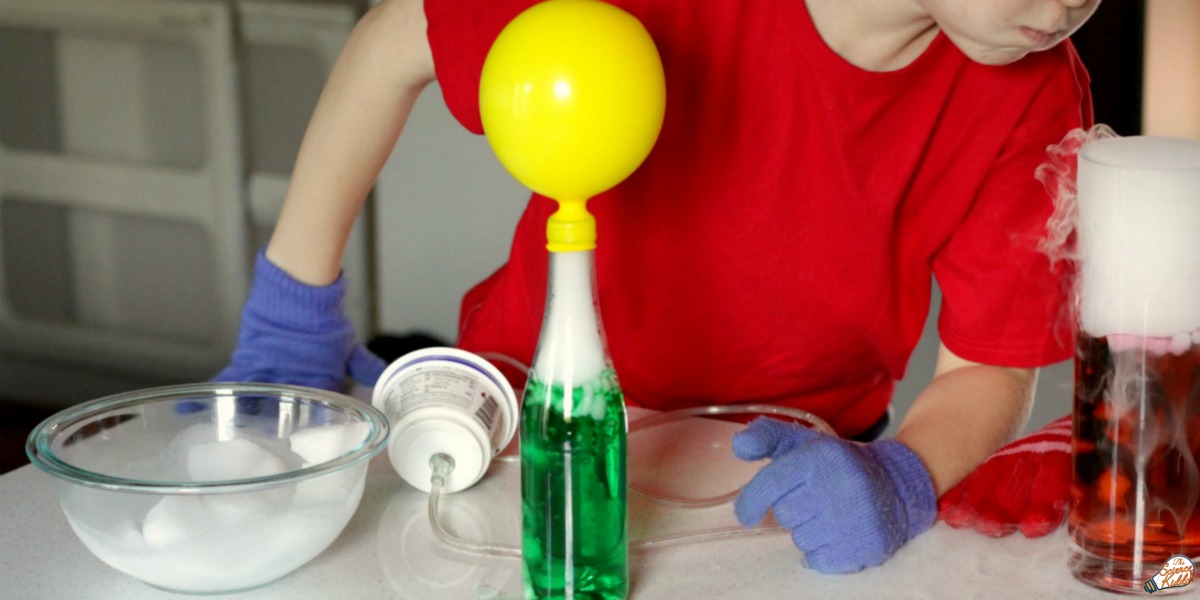
While wearing gloves, I placed a few pieces of dry ice into the bottle of water. The kids never get tired of watching the dry ice fog spill out of the bottle while the dry ice bubbles inside. It’s always amazing!
My son stretched an empty balloon across the mouth of the bottle. Within seconds the balloon started to inflate, as if by magic!
We used 12 inch party balloons so we were able to leave the balloon on the bottle until the dry ice sublimed completely away. If you are using a smaller balloon or more dry ice, you may want to be careful not to overfill the balloon. There is a chance it could get overfilled with carbon dioxide and pop!
Since we had tons of dry ice and several balloons we used them to inflate a balloon with dry ice over and over again. We never grew tired of watching the balloon get bigger and bigger!
Dry Ice Science
Dry ice is solid carbon dioxide. At temperatures above -109 degrees Fahrenheit, or -78.5 degrees Celsius, dry ice turns into carbon dioxide gas. (This is the same gas you exhale as you breathe.) The transition from a solid to a gas is called sublimation.
Water isn’t strictly necessary to make the dry ice sublime. However, immersing the dry ice in warm water makes it sublime more quickly and produces a really cool cloud of white vapor.
The fog bubbling out of the dry ice water is a mixture of condensed water vapor and carbon dioxide gas. It’s essentially the same stuff clouds in the sky are made of!
The idea behind this dry ice project is to trap the fog inside the balloon, forcing it to inflate. The more dry ice there is in the bottle, the bigger the balloon will inflate!
An interesting dry ice experiment that incorporates math and science together would be to weigh the solid dry ice before placing it in the water and then weigh the inflated balloon once all the dry ice sublimed into it. Do you think it would weigh the same? More? Less?













I’m interested in taking ur balloon idea one step further. My idea is to place the filled balloon inside the hollow of a cake. When it’s sliced, the knife will prick the balloon + smoke is emitted.
How long will the balloon stay inflated?
Thanks for any help you can provide!
Wow, that’s a really cool idea! I don’t understand exactly what you have in mind with putting the balloon inside a cake, but I think it sounds really cool. The balloon should stay inflated for at least a few hours, but over time the air does start to seep out. If I were you I would experiment with filling a balloon with dry ice a few times before inserting it into the cake just to make sure it works the way you want it to and it stays inflated long enough.
It’s look like some thing interesting, I want do it but I didn’t find dry ice.